The Disease
Small doses of endotoxin induce in mice acute inflammatory responses similar to those occuring during the early stages of septic shock. Within hours following the administration of endotoxin, changes occur in organ functions and inflammatory cytokines & chemokines like TNF and IL6 are released.
Co administration of D-galactosamine N (GalN), results in the development of acute hepatic failure.
Treatments
Blockade of TNF or TNFR1 results in the reduction of inflammatory responses and the rescue of mice from hepatic failure.
Our Preclinical Testing Tools
LPS acute sepsis (Cytokine release)
This animal model provides a unique fast screening platform for the evaluation of the efficacy of anti-inflammatory drugs that counteract the LPS-induced responses.
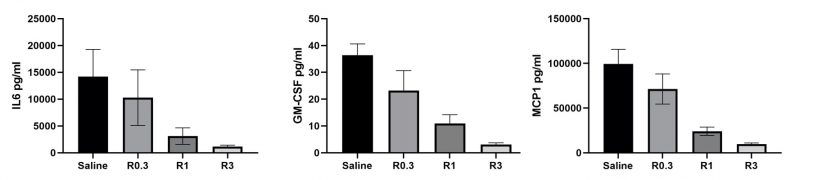
To the right, Remicade counteracts LPS induced IL6, GMCSF and MCP1 levels in hTNF transgenic mice, in a dose dependent manner.
GaIN-LPS hepatic failure
The GalN-LPS model of hepatic failure can be induced in either wild type, hTNF transgenic mice or hTNFR1 knockin mice and it offers a fast and highly sensitive screening platform for the efficacy evaluation of anti-hTNF , anti-hTNFR1 therapeutics and small molecules.
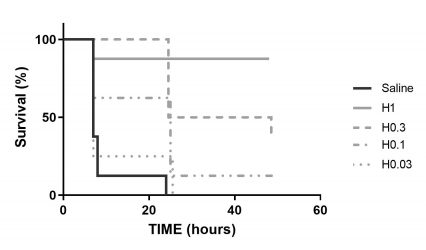
To the right, Humira rescues hTNF transgenic mice from hepatic failure in a dose dependent manner.
Drug Efficacy Evaluation Platforms
Mice receive intraperitoneal injections of LPS alone or in combination to D-galactosamine (GalN). In the absence of efficient treatment high levels of proinflammatory cytokines are released, while in the presence of GalN, lethality reaches 100% within a few hours. Efficient treatment can lower the levels of secreted cytokines and can eliminate lethality in a dose dependent manner.
Read-Out Parameters
Evaluation of serum levels of cytokines and chemokines, including TNF, IL6, MCP1, CXCL1
Survival (for GalN-LPS experiments)
ALT levels (for GaIN-LPS experiments)
Competitive Advantage
Both models offer fast and highly sensitive screening platforms of anti-TNF and anti-TNFR1 therapeutics at all stages of their development. Efficacy of dozens of compounds can be screened in one day. Sensitivity is high so that even low doses of anti-TNF therapeutics offer protection.