- Spontaneous Model(s) hTNFTg, TNF ΔARE/+
- Induced Model CAIA
- Performed on TNF, TNFR1
- Readouts In vivo scoring, Histopathology

The Disease
Rheumatoid arthritis (RA) is a disease characterized by chronic inflammation of the joints associated with bone and cartilage destruction. RA is a severe burden to patients leading to disability, pain, severe impairment of quality of life, and even life threatening conditions resulting in significantly enhanced mortality.
The hyperplastic synovium and infiltrating inflammatory cells produce proinflammatory cytokines such as TNF-alpha and destructive enzymes that lead to cartilage destruction and bone erosion.
Treatments
TNF-alpha inhibitors dominate in the clinic as first-line biological agents. The major players in the arthritis drug market include Remicade® (infliximab), Humira® (adalimumab), Enbrel® (etanercept), Cimzia® (certolizumab pegol) and Simponi® (golimumab), while biosimilars of the above as well as new drugs with novel mechanisms of action are under development.
Our Preclinical Testing Tools
Biomedcode’s unique animal models of human TNF-depended arthritis have historically offered the first proof of concept for the central role of TNF in the development of human arthritis. Our rich collection of arthritis mouse models combined with standardized protocols allow the accurate preclinical efficacy evaluation of anti-arthritic pharmaceuticals and offer an ideal platform for establishing proof of concept for novel technological advances.
Human TNF driven polyarthritis (Tg197)
A humanized mouse model that overexpresses human TNF and develops spontaneously chronic arthritis pathology closely resembling human rheumatoid arthritis. The Tg197 mouse model offered the first proof of concept for the central role of TNF in the development of human arthritis and up today it has been used for the efficacy evaluation of numerous anti-hTNF therapeutics.
Tg197 model crossed to hTNFR1KI mice yields Tg197hTNFR1KI, a double humanized arthritis model that offers the opportunity to test anti-hTNFR1 efficacy in direct comparison to that of anti-hTNF biologics or to test the effect of anti-hTNF biologics in a humanized TNF-TNFR1 system.
Human tmTNF driven arthritis (Tg5453)
A humanized transgenic mouse with deregulated expression of the transmembrane form of human TNF that spontaneously develops early onset severe arthritis pathology. This model offers a unique tool for the assessment of the efficacy of anti-hTNF agents in the blockade of the transmembrane form of human TNF.
Human TNF driven slow progressing arthritis (Tg3647)
A humanized TNF transgenic mouse overexpressing human TNF that spontaneously develop late onset, slow progression, long lasting arthritis pathology. This model is ideal for the testing of therapeutics that require long duration administration and observation.
Mouse TNF driven arthritis and IBD (TNF ΔARE/+)
A mouse mutant with deregulated TNF expression resulting in the spontaneous development of arthritis together with Crohn’s –like intestinal inflammation. An ideal model for the evaluation of therapeutics in a complex pathology model.
TNFΔΑRE model crossed to hTNFR1KI mice yields TNFΔAREhTNFR1KI, a model that offers the opportunity to test anti-hTNFR1 therapeutic efficacy in both arthritis and Crohn’s like intestinal inflammation.
Collagen Antibody Induced Arthritis (CAIA) in wt and hTNF transgenic mice
A highly reproducible induced mouse model of rheumatoid arthritis offering the advantage of a short duration experimental protocol that facilitates the fast and efficient evaluation of candidate therapeutics, especially useful when small amounts of compounds are available.
CAIA induction in the normally regulated Tg1278 human TNF transgenic mice results in the generation of a sensitive and highly reproducible arthritis mouse model ideal for the fast efficacy evaluation of anti-human TNF therapeutics.
Arthritis Evaluation Platforms
Mice can be treated either prophylactically -at disease onset- or therapeutically –at established pathology- with protocols standardized for the evaluation of a variety of therapeutics. Disease progress and severity are assessed regularly using validated readouts with standardized procedures.
Read-Out Parameters
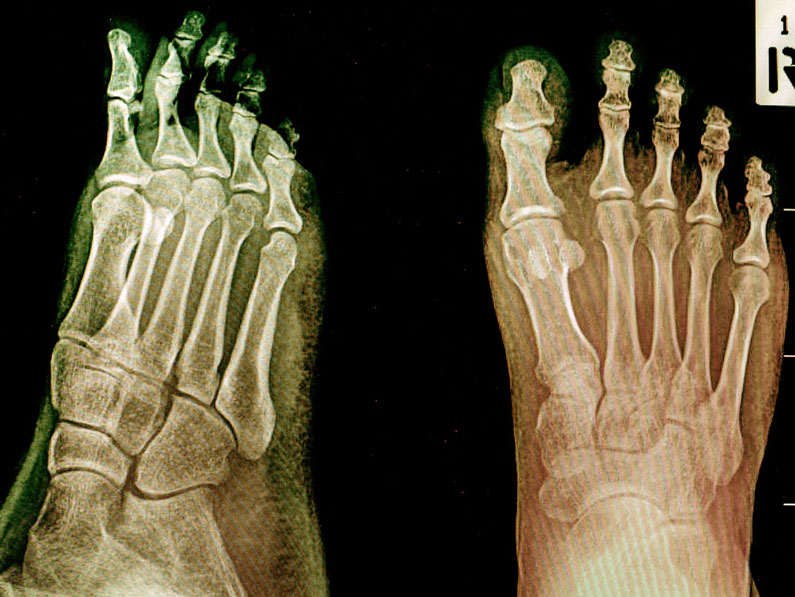
In vivo arthritis scoring
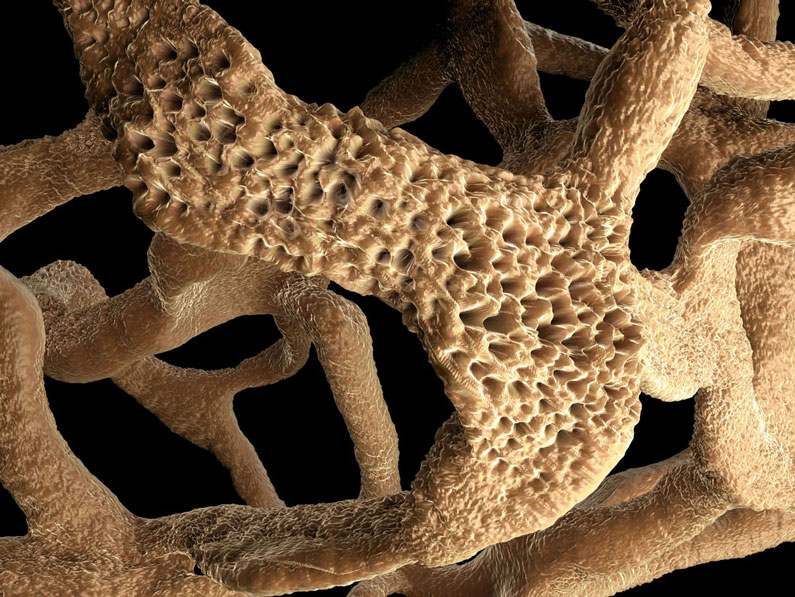
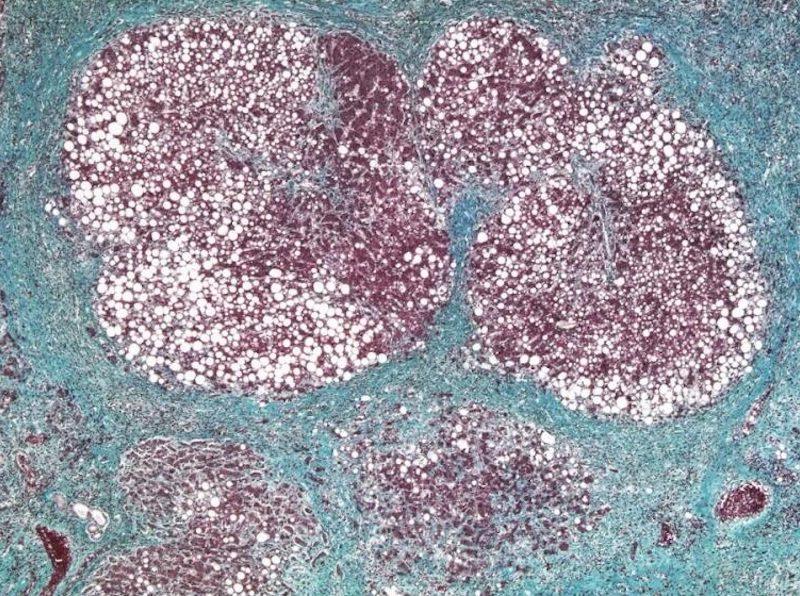
Histopathological evaluation of joint sections
Inflammatory cytokine serum levels measurements
Competitive Advantage
Biomedcode’s unique collection of mouse models of arthritis in combination with the standardized evaluation procedures developed over years of experience in the preclinical efficacy evaluation of arthritis therapeutics, offers the opportunity for reliable evaluation of the efficacy of anti-human TNF biologics biosimilars and small molecule inhibitors of pathology related pathways.