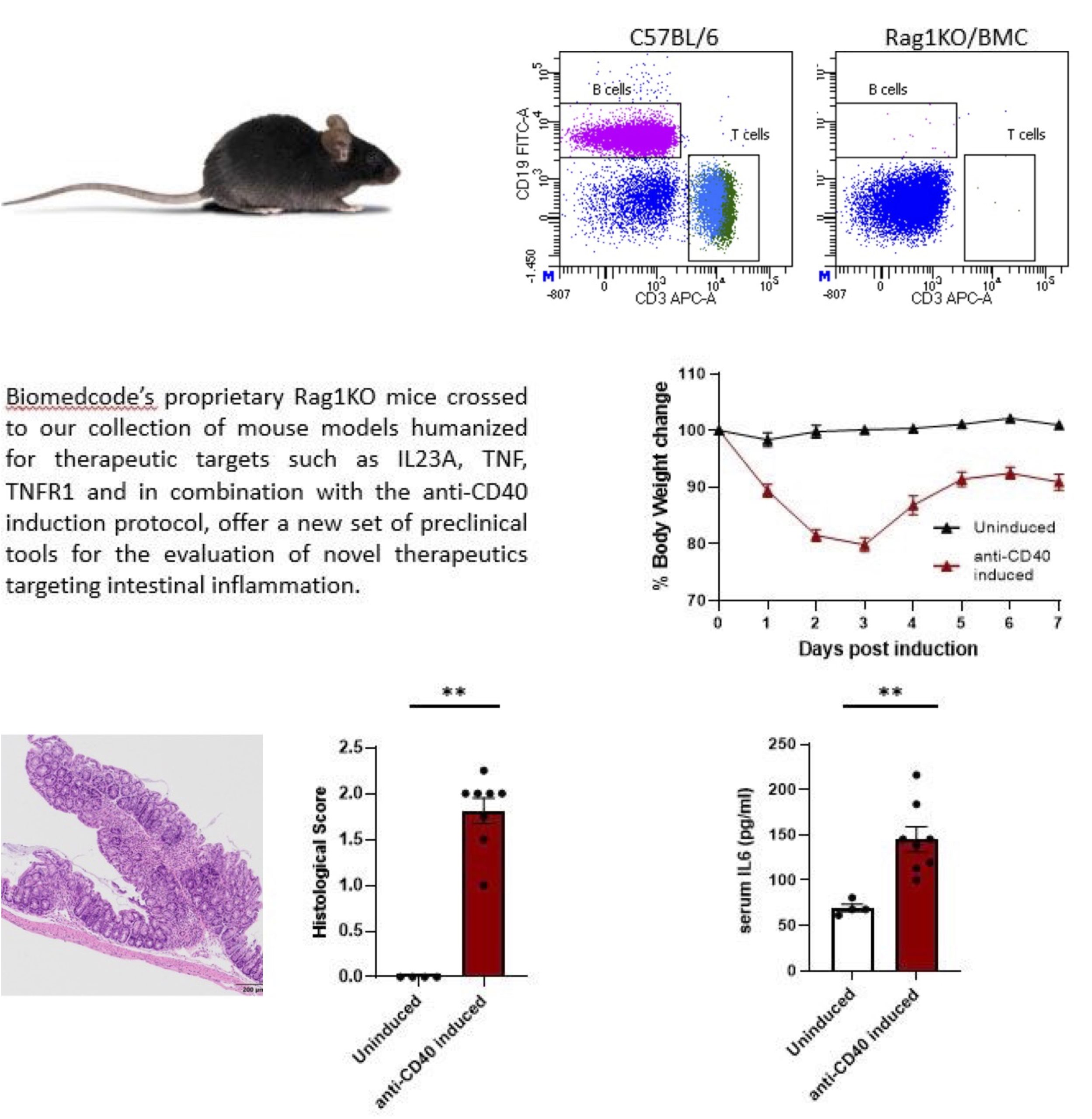
Scleroderma or systemic sclerosis is a rare connective tissue disorder with complex pathogenesis. Scleroderma can be divided in localized scleroderma primarily affecting the skin and subcutaneous tissue, whereas systemic sclerosis is associated with systemic manifestations and involvement of multiple organ systems, including kidney, lung, heart, gastrointestinal tract and more.
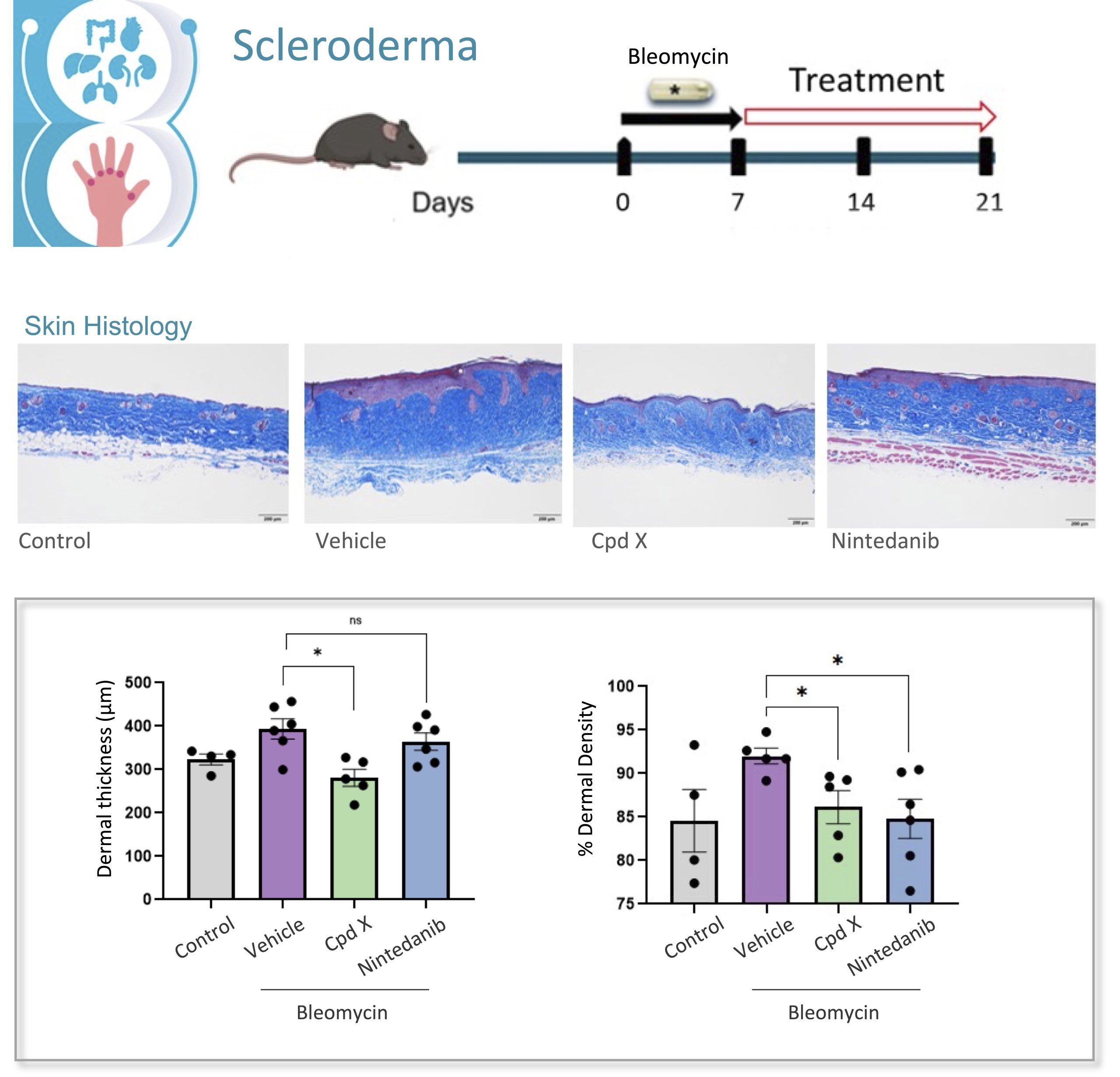
To support the development of antifibrotic therapies that will be effective in the treatment of scleroderma we have developed and standardized the bleomycin induced scleroderma animal model, that reproduces the pathology manifestations and is widely use in preclinical research.
The scleroderma model induced by mini osmotic pump released bleomycin integrates comprehensive histopathological and molecular readouts of fibrosis and inflammation, providing a well-established and highly translational in vivo preclinical platform for evaluating novel therapeutics targeting fibrosis.



INFRAFRONTIER has achieved two major milestones in the first half of 2024, with two collaborative projects being funded by the European Commission INFRAPLUS and PRIMTECH3R
Biomedcode participates in both projects together with other partners and collaborators:
Helmholtz Munich – ERA-LEARN – Institut Clinique De La Souris ICS – CIPHE – Centre d’Immunophénomique – University of Oulu – Karolinska Institutet – CSIC – Centro Nacional de Biotecnología (CNB-CSIC) – Consiglio Nazionale delle Ricerche – Vetmeduni – BSRC Alexander Fleming – Universitat Autònoma de Barcelona – UK Research and Innovation – Mary Lyon Centre at MRC Harwell – MRC National Mouse Genetics Network – Biomedcode – AKITA, by Finnadvance – CNRS – Calouste Gulbenkian Foundation Instituto Gulbenkian de Ciencia – The Netherlands Cancer Institute – Antoni van Leeuwenhoek – The Hospital for Sick Children – Institute of Molecular Genetics of the Czech Academy of Sciences – Czech Centre for Phenogenomics – UMCG research – EMBL – INSERM and Aarhus University