

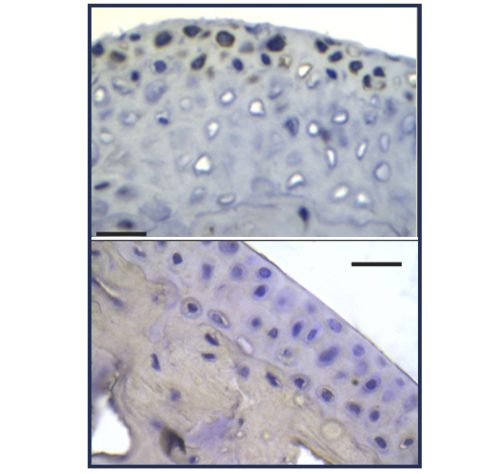
Extending on our previous work on the treatment of arthritis with combination therapies of dasatinib with subtherapeutic doses of anti-TNF biologics, we explore here in this collaborative research project the interplay of inflammation and senescence in the context of combination treatments.
Published in Mechanisms of Ageing and Development 2023, 214, 111856.
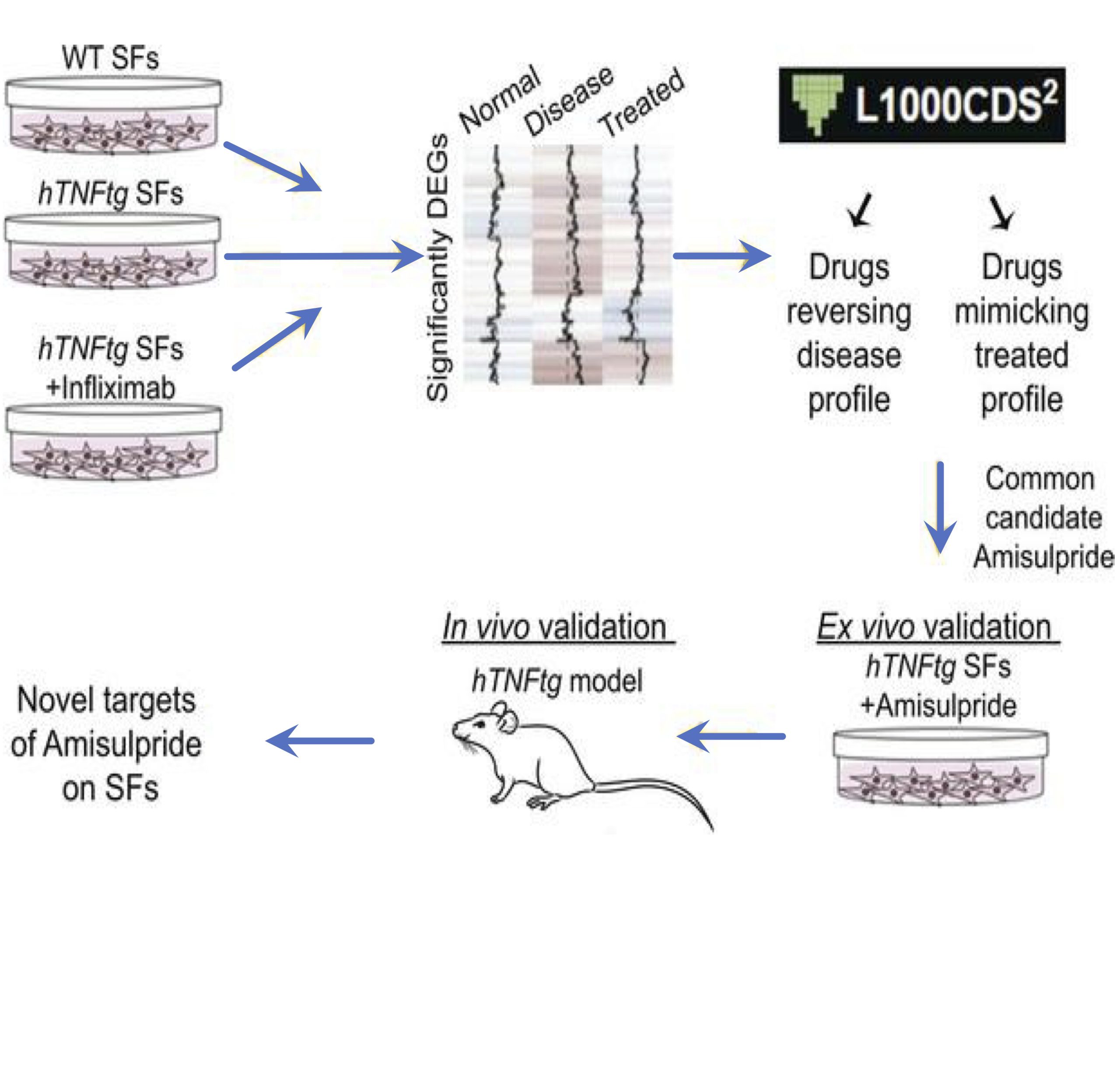
In the frame of a collaborative drug development project scientists from Biomedcode and BSRC Al. Fleming using bioinformatics tools, have repurposed the neuroleptic drug amisulpride for the reversal of the pathogenic expression signature of synovial fibroblasts and the treatment of arthritis pathology.
Published in JCI Insight 2023 May 8;8(9):e165024. doi: 10.1172/jci.insight.165024.
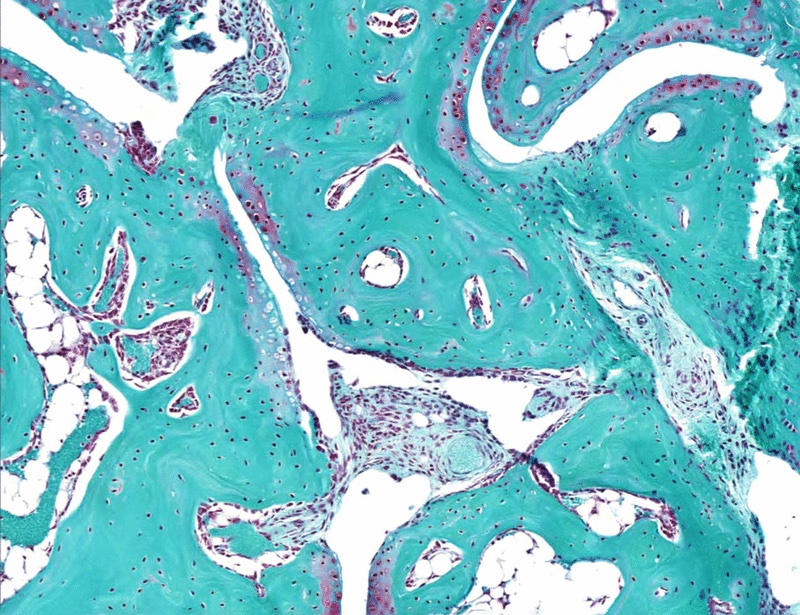
In this paper the anti-TNF/IL6 bispecific nanobody is tested on the Tg197 arthritis model.
Published in Science Translational Medicine 2023 Feb;15(681):eabq4419. doi: 10.1126/scitranslmed.abq4419.
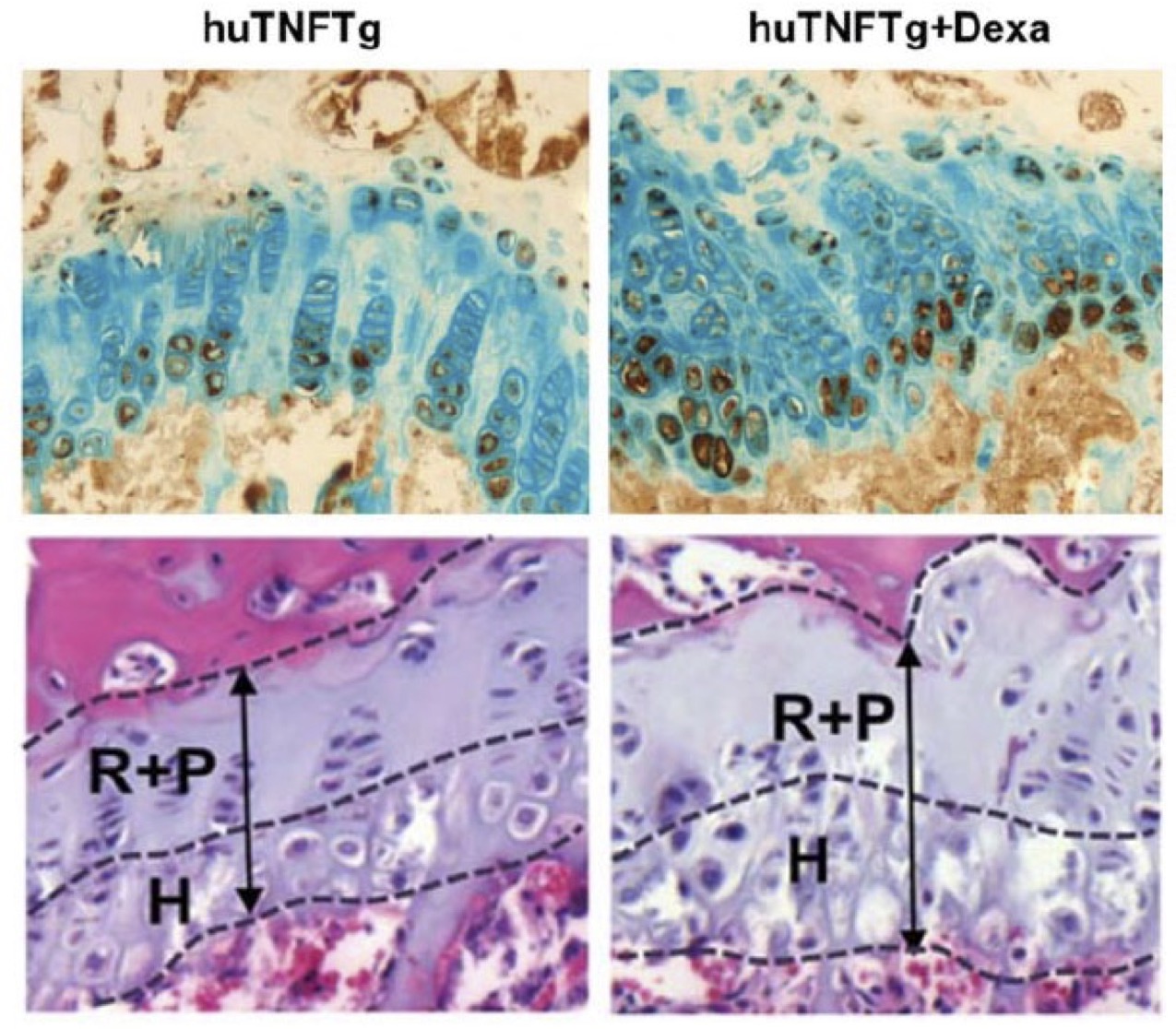
Published in Scientific Reports 2022 Oct 28;12(1):18189. doi: 10.1038/s41598-022-22734-8
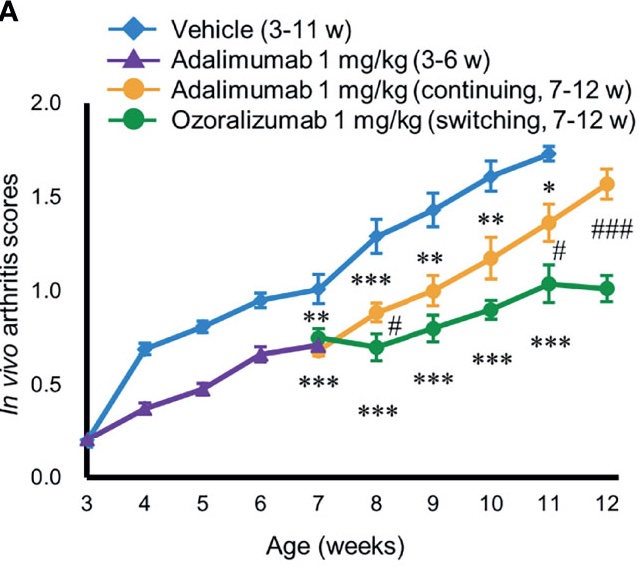
In this recent paper, Ishiwatari-Ogata et all, using the Tg197 mouse model of arthritis, show that Ozoralizumab, a trivalent, bispecific NANOBODY compound offers anti-arthritis efficacy even in the secondary-failure phase.
Published in Frontiers in Immunology 2022 Feb 22;13:853008. doi: 10.3389/fimmu.2022.853008.
Read More
Our paper in Journal of Translational Medicine shows that combination of dasatinib and other kinase inhibitors with a subtherapeutic anti‐hTNF dose effectively treats arthritis pathology.
Published in Journal of Translational Medicine 2021 Apr 23;19(1):165. doi: 10.1186/s12967-021-02764-y.

Biomedcode has been recognized by the Pharma Tech Outlook magazine as one of the top 10 CROs of the year 2020. An interview with Biomedcode’s CEO and CSO is published in this month’s special edition on Europe’s CROs.


Biomedcode is excited to participate as one of the 63 European companies that have been selected to join the EUGATEWAY Healthcare and Medical Technologies Business Mission to Singapore on 8-11 December 2020. Due to the COVID-19 pandemic this will be a virtual event and we look forward to discussing with potential new partners and clients about future collaborations!
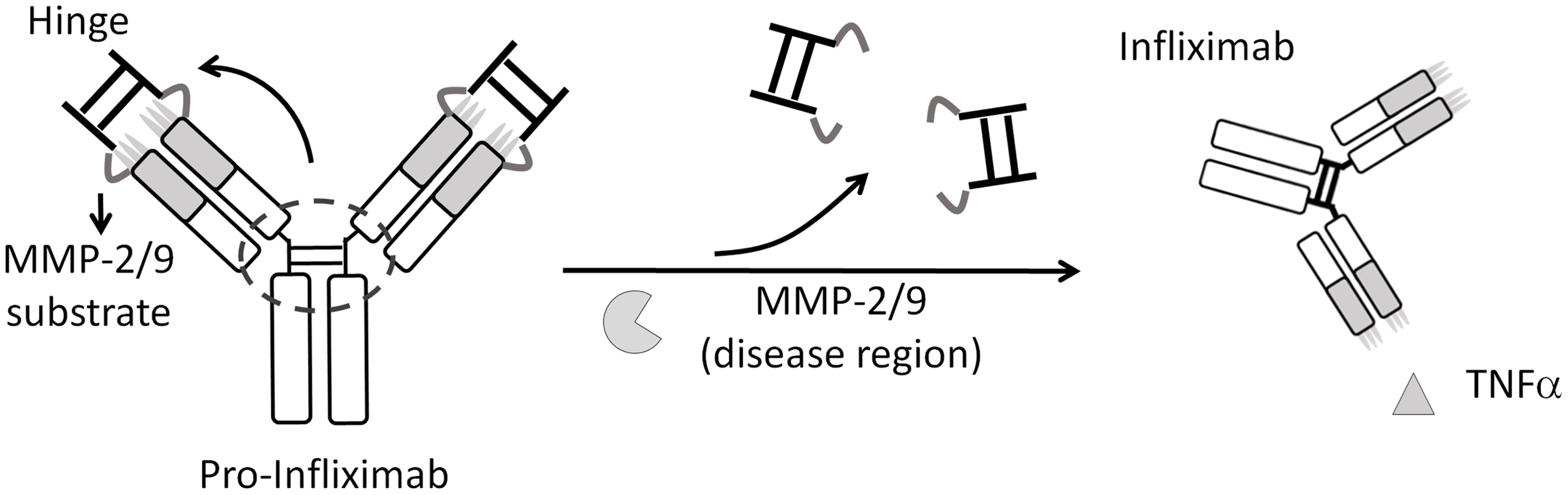
Pro-Infliximab provided equivalent therapeutic efficacy to Infliximab while maintaining mouse immunity against Listeria infection, leading to a significantly higher survival rate (71%) than that of the Infliximab treatment group (0%).
Published in PLoS Biology 2019 Jun 13;17(6):e3000286. doi: 10.1371/journal.pbio.3000286.

INFRAFRONTIER has achieved two major milestones in the first half of 2024, with two collaborative projects being funded by the European Commission INFRAPLUS and PRIMTECH3R
Biomedcode participates in both projects together with other partners and collaborators:
Helmholtz Munich – ERA-LEARN – Institut Clinique De La Souris ICS – CIPHE – Centre d’Immunophénomique – University of Oulu – Karolinska Institutet – CSIC – Centro Nacional de Biotecnología (CNB-CSIC) – Consiglio Nazionale delle Ricerche – Vetmeduni – BSRC Alexander Fleming – Universitat Autònoma de Barcelona – UK Research and Innovation – Mary Lyon Centre at MRC Harwell – MRC National Mouse Genetics Network – Biomedcode – AKITA, by Finnadvance – CNRS – Calouste Gulbenkian Foundation Instituto Gulbenkian de Ciencia – The Netherlands Cancer Institute – Antoni van Leeuwenhoek – The Hospital for Sick Children – Institute of Molecular Genetics of the Czech Academy of Sciences – Czech Centre for Phenogenomics – UMCG research – EMBL – INSERM and Aarhus University